In biological systems, regulatory proteins are essential for a wide range of cellular activities based on interactions with the local environment. Therefore, detailed knowledge of protein−ligand (or protein−protein) interaction is required to understand biological processes. A profound knowledge of these interactions is a crucial prerequisite to translate our research into drug design. Using solution-state NMR spectroscopy and other biophysical methods such as CD, UV/Vis and fluorescence spectroscopy, we analyze structures of small molecules and proteins, as well as protein-ligand interaction and their dynamics, e.g., of cytokines /proteases in complex with their ligands.
Standard techniques in our lab include:
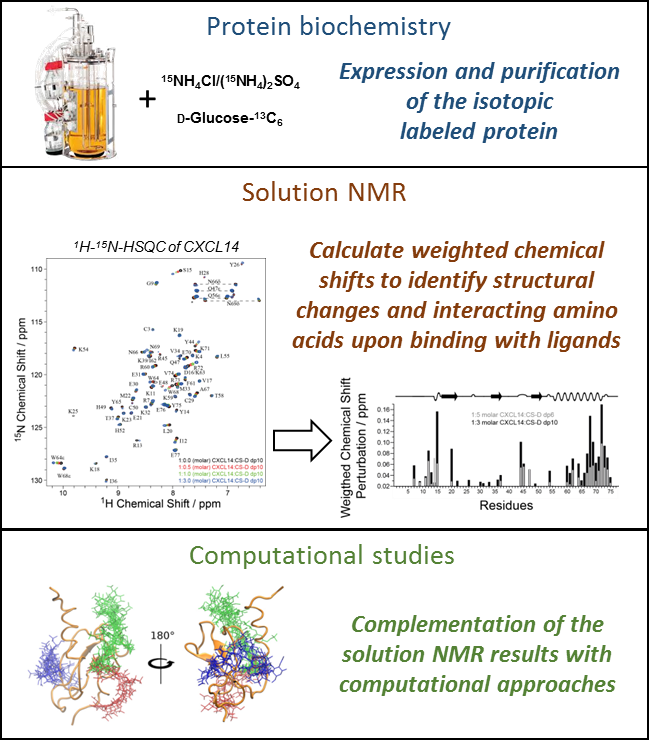
- Recombinant Expression, refolding and purification of isotopically labeled (e.g. 13C, 15N) protein
- several solution NMR methods (e.g. 1H-15N-HSQC titration, pseudo contact shifts (PCS), saturation transfer difference (STD), assignment of proteins, … )
Key Publications:
- A. Penk, L. Baumann, D. Huster, S. A. Samsonov. NMR and molecular modeling reveal specificity of the interactions between CXCL14 and glycosaminoglycans. Glycobiol. 2019, 29, 715−725.
- G. Künze, S. Köhling, A. Vogel, J. Rademann, D. Huster. NMR characterization of the binding properties and conformation of glycosaminoglycans interacting with interleukin-10. J Biol Chem. 2016, 219, 3100–3113.
- A. Pichert, S. A. Samsonov, S. Theisgen, L. Thomas, L. Baumann, J. Schiller, A. Beck-Sickinger, D. Huster, M. T. Pisabarro. Characterization of the interaction of interleukin-8 with hyaluronan, chondroitin sulfate, dermatan sulfate and their sulfated derivatives by spectroscopy and molecular modeling. Glycobiol. 2012, 22, 134–145.
Team:
Dr. Anja Penk
Dr. Benedikt Schwarze
Christian Schulze
Collaborations:
- Dr. Sergey A. Samsonov, Faculty of Chemistry, University of Gdánsk, Poland
- Prof. Dr. Jörg Rademann, Faculty of Pharmacy, Freie Universität Berlin
- Dr. Sven Rothemund, Core Unit Peptide, Leipzig University
- Prof. Dr. Annette G. Beck-Sickinger, Institute of Biochemistry, Leipzig University
Funding:
Transregio-SFB TRR 67, project A6
DFG project HU 720-19-1